
SkinBusiness offers a complete set of components and training to enable a practitioner to separate regenerative cells from lipoaspirate without the use of enzymes. This information and technology is provided by SkinBusiness solely as an equipment option and does not promote any specific Regenerative Cell Therapies*.
- Description
- Technologies
- More Information
It has been discovered that adipose tissue contains high concentrations of regenerative cells which can be easily extracted for use in other applications.
SkinBusiness can provide equipment and training on a technique that delivers an extremely high quantity of regenerative cells without the use of enzymes, and only requires about 35 to 45 minutes to perform.
This technique requires very little manipulation of the adipose tissue and does not involve the use of enzymes. Due to these facts, the procedures physicians choose to perform involving the regenerative cells produced by this technology may be considered less controversial than other current cell processing techniques or technologies.
* Regenerative Cell Therapies are not currently approved by the FDA.
 Nutational Infrasonic Liposculpture System (aka Tickle Lipo)
Nutational Infrasonic Liposculpture System (aka Tickle Lipo)
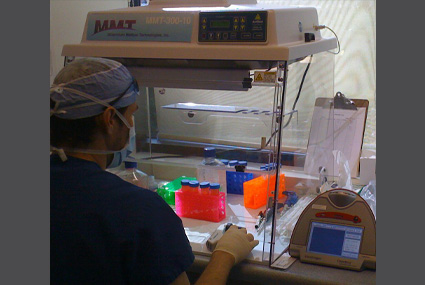
 MMT Regenerative Cell Processing Technology and Training
MMT Regenerative Cell Processing Technology and Training
 MMT Fat Collection System
MMT Fat Collection System
The collection and processing of regenerative cells is becoming one of the fastest growing segments of the liposuction marketplace. Since collecting stem cells from bone marrow can be an uncomfortable experience for the patient, the collection of ADRC's (Adipose Derived Regenerative Cells) is a preferred choice among patients and physicians. Additionally, bone marrow extractions collect anywhere from 3 million to 5 million regenerative cells, while ADRC's collected during a standard Tickle Lipo procedure can be upwards of 100 to 200 million per 50 cc sample (dependent on patient age, health, and other factors). There are restrictions mandated by the FDA if regenerative cells are extracted with more than minor manipulation, which includes processes that involve the use of enzymes. Practitioners must be aware of the rules, written or implied, before presenting any treatment to their patients.
* Regenerative Cell Therapies are not currently approved by the FDA.